CRM for sanitary and technical food laboratory
Software for automation of the sanitary and technical food laboratory. STPL conducts physico-chemical and microbiological studies for compliance with safety and quality requirements. STPL is accredited in the accreditation system.
The software has a system of reference books
- Counterparties
- Price list
- Types of test objects according to the FSA nomenclature
- Types and groups of test objects with identification features
- ND, etc., etc. on trial (sample)
- ND, etc., etc. for a sample (sample) to groups of test objects with indication of indicators and their permissible levels
- Test performance
- Group of indicators
- ND on test methods
- Types of tests
- Testing (research) departments of the
- Units of measurement
- Customer type
- Types of packaging
- Transportation methods
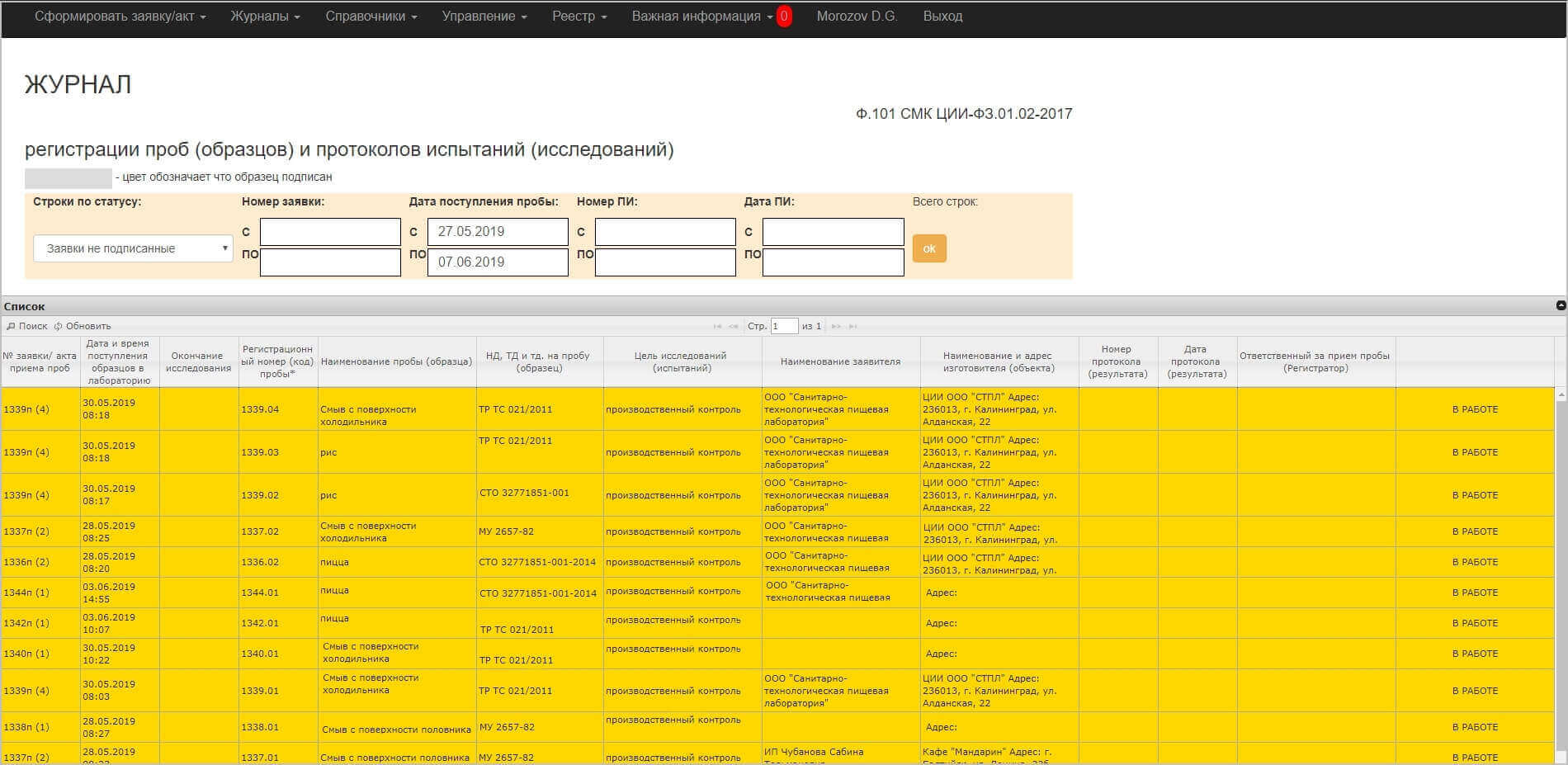
As well as three main magazines
- Log of registration of samples (samples) and issuance of results
- Log of registration of samples (samples) of physico-chemical tests
- Journal of registration of samples (samples) of microbiological tests
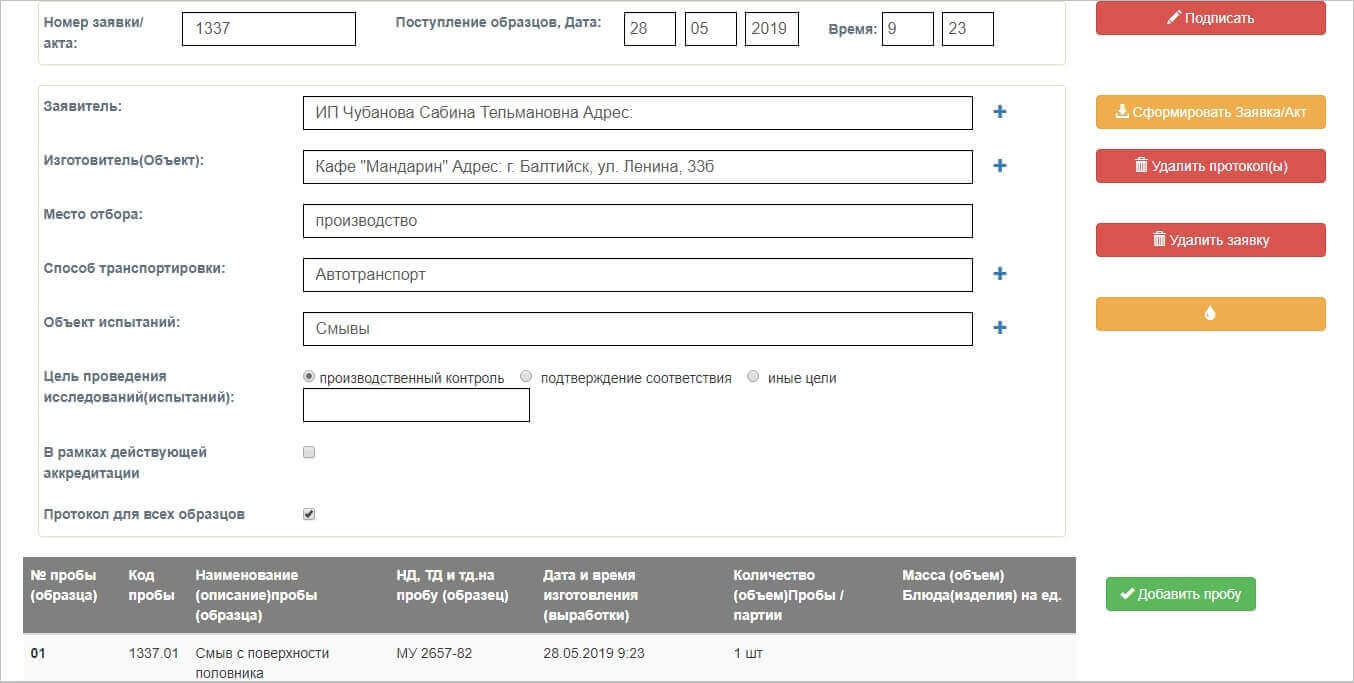
To make the selected samples/samples from the research objects, the registrar creates an Act/ application, which consists of the following parts
Basic information about the Counterparty, the object and sampling.
Next, samples/samples are created for research. A given unique code is generated for each sample, by which it can be identified in any external documents of the system. Each sample is assigned a specific list of the studied indicators according to the Regulatory Documentation, according to which the laboratory assistant will conduct research in the future And additional information to the Act / application is indicated at the end
After all the data is filled in, the Certificate / Application is signed by the registrar and all samples are sent to the journals for laboratory assistants for physico-chemical or microbiological studies, depending on the studied parameters of this sample. The laboratory assistant sees the samples, but does not see to which organization this sample belongs – the principle of equality and impartiality is observed. And then conducts research and records the results of research in the necessary fields. Having formed a complete table with the data, the laboratory assistant signs the study for this sample – as if completing all the work on it. A Protocol is formed based on the results of the signature.
According to the results of all studies, an upload is formed to the Federal Accreditation Service. From the platform, you can upload an Act/application, research protocols, and Journals in xls format. Generate invoices and certificates of work performed for billing to counterparties. Generate and upload contracts for Counterparties, as well as additional agreements, and a Price List separately for each counterparty. Upload selected metrics. Documents in the FSA.
The software has an additional Important Information section – this is a section with documents for employees - Internal and External regulatory documents. Register of invoices and certificates of work performed. A register of all documents that have ever been generated in the Web Service. Passport of the organization that carries out research. A subsection of non-accredited (production control) studies working simultaneously with accredited ones and the ability to hide (disable) it.
The platform has a built-in system of registration, authorization, user roles and management of user profiles and data. The web service allows you to register new users and add them to pre-created roles (Administrator, registrar, technologist and laboratory assistant). Each role has its own data access rights. The administrator can work in the system without restrictions, the registrar has the right to work with the database of clients, contracts and invoices, and acts of work performed, with acts of selection, directions for research and printing of relevant documents (contract, invoices, price list, journals (5 pieces) and reports (5 pieces)) The registrar sees only his part of the work (everything except access control to the system). The technologist has rights and functionality such as a registrar. Laboratory assistant - receives the data necessary for the study, corrects and enters the research data (work logs) and results, works with the research results and can print out the relevant documents (direction, research report for the period) and sees only his part of the work.






Opportunities
- More than 15 reference books
- The work of laboratories in several areas
- Document flow
- Journals by type of research
- Managing users and roles
- Formation of an upload to the FSA registry